
It is possible to be quantitative about the probability of finding a “billiard-ball” electron at a given location, however. 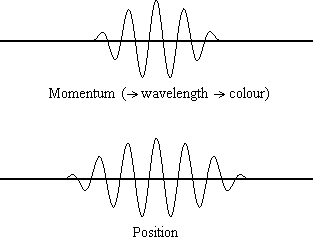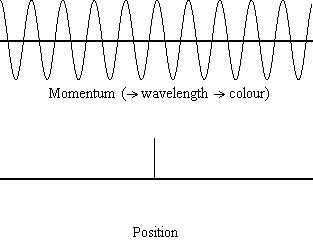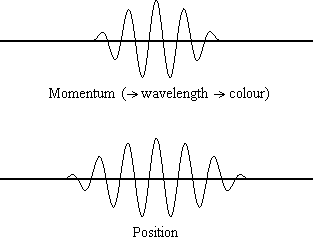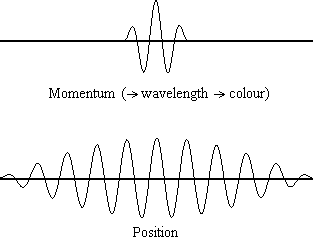
There is a finite probability that it was at the other place to begin with! Since it is impossible to know precisely where the electron is at a given instant, the question, “How does it get from one place to another?” is pointless. It will be possible to talk about the probability that the electron is at a specific location, but there will also be some probability of finding it somewhere else in the box. This means that determination of the electron’s position will be very inexact. (4) in Wave Mechanics assigns exact values of kinetic energy to the electron in the box, the speed can be calculated accurately. Since kinetic energy depends on speed (½ mu 2) and Eq. 1 a in Wave Mechanics), it will be impossible to determine both the electron’s exact position in the box and its exact speed. So to pinpoint the location of an electron, we must change its momentum.Īccording to the uncertainty principle, even if we draw an analogy between the electron in a box and a billiard ball (Fig. Another more relevant example is that in order to detect an electron, a photon must first interact with it, and this interaction will change the path of that electron. For example, a thermometer must absorb some heat to record a temperature, and therefore must reduce the temperature of the matter that is being measured. The "observer effect" is the change in a phenomenon that the act of observation will make on the phenomenon. The uncertainty principle is related to the observer effect, with which it is often conflated. We can think and control motions pretty much without error, so the Heisenberg uncertainty argument for free will (or control of our actions by a god) need to address this issue. If this were literally true, our thoughts and muscular control would be much more random than they are, due to the uncertainty of position of the neurotransmitter. Since this is much larger than the dimensions of a molecule (say 1000 picometers, ~10 -9 m), the uncertainty position of the molecule, and the probability that it will dock incorrectly on the receptor nerve, is pretty high.īut the velocity of the molecule, and chemical forces that may more precisely control its velocity and thus docking position, are not known, as should be obvious from this result.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
